U.S. (WAND) - Aurobindo Pharma USA, Inc. is recalling a large supply of blood pressure medication due to a impurity that may cause cancer.
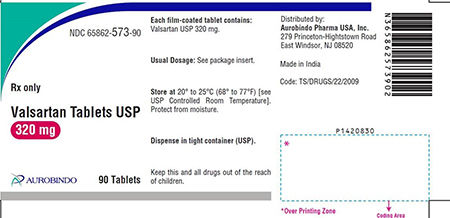
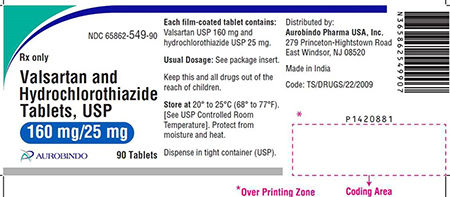
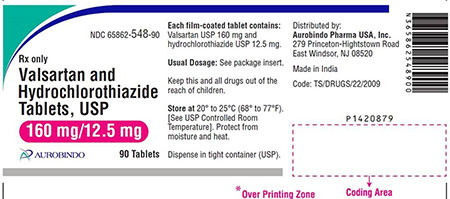
The impurity detected in the finished drug product is N-nitrosodiethylamine (NDEA), which is a substance that occurs naturally in certain foods, drinking water, air pollution, and industrial processes, and has been classified as a probable human carcinogen as per International Agency for Research on cancer classification. To date, Aurobindo Pharma USA, Inc. has not received any reports of adverse events related to this recall.
Amlodipine Valsartan Tablets USP, Valsartan HCTZ Tablets, USP and Valsartan Tablets USP are used to control high blood pressure and as treatment for heart failure. Patients should contact their pharmacist or physician who can advise them about an alternative treatment prior to returning their medication.
Consumers should also contact their physician or healthcare provider if they have experienced any problems that may be related to taking or using this drug product.
Any general questions regarding the return of this product please contact Inmar\CLS-Medturn at 1-877-208-2407 or email rxrecalls@inmar.com (live calls received 9 am -5:00 pm Eastern Time).